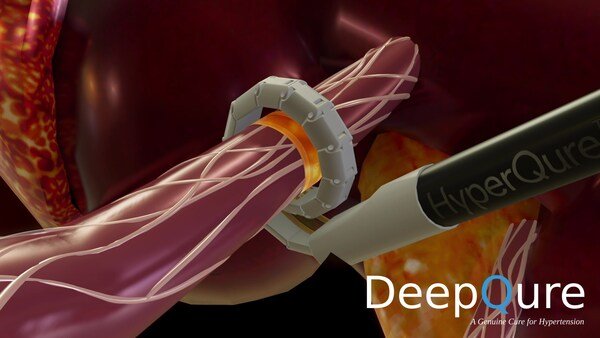
DeepQure, a Seoul based medical device company, announced initiation of its Early Feasibility Study (EFS) for the HyperQure system following FDA IDE approval. HyperQure is the world's first extravascular RDN medical device for the treatment of resistant hypertension.
With this approval, the company will commence the Early Feasibility Study to prove the safety and efficacy of HyperQure in 15 patients with resistant hypertension. The clinical trial will be conducted in a prospective, multicenter, single-arm, open-label design at major U.S. university hospitals, including Stanford University, Mayo Clinic, Emory University, University of Arizona and the University of California, Irvine.
HyperQure system consists of an energy generator and a laparoscopic instrument that delivers RF energy for ablation directly to the sympathetic nerves around the renal artery by wrapping the renal artery 360 degrees from outside of the vessel. This mechanism allows full denervation of renal sympathetic nerves without damaging the vascular endothelium which has proven to be a challenge for intravascular (catheter) systems.